Coming Off Wegovy and Ozempic: The Evidence-Backed Semaglutide Off-Ramp
Summary
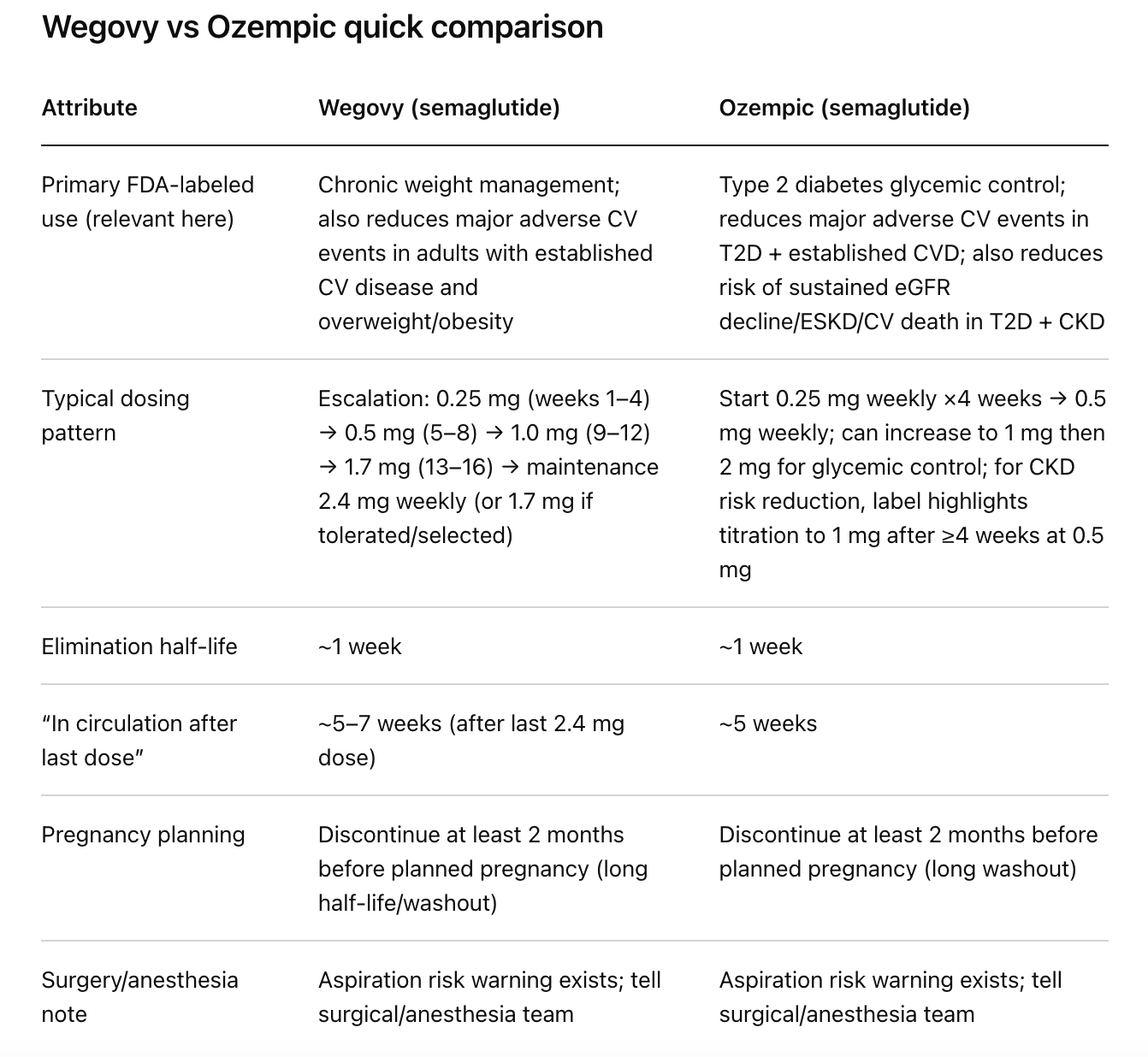
Stopping semaglutide (whether you were taking it as Wegovy or Ozempic) should be considered a biology handoff. Semaglutide has an elimination half-life of about one week, and it can remain in circulation for about 5–7 weeks after the last 2.4 mg Wegovy dose and about 5 weeks after the last Ozempic dose. That long “tail” is why many people feel okay at first, then notice hunger and weight rebound ramping up later. [1]
The most cited discontinuation data point is real: in the STEP 1 trial extension, one year after stopping semaglutide 2.4 mg, participants regained about two‑thirds of their prior weight loss, and cardiometabolic improvements drifted back toward baseline. [2] A separate withdrawal design (STEP 4) showed the same directional truth more cleanly: people who continued semaglutide kept losing weight, while those who switched to placebo regained weight during the randomised phase. [3]
A 2026 systematic review/meta-analysis in The BMJ puts “rebound” into a broader frame: after stopping weight management medications, average regain was estimated at ~0.4 kg/month, with faster regain after newer incretin drugs, and cardiometabolic markers projected to return toward baseline within roughly 1.4 years after cessation (based on modelled trends). Here’s the contrarian kicker: the review found no evidence that more intense behavioural support during treatment (or behavioural support after stopping) meaningfully changed the rate of regain suggesting that “just build habits while you’re on it” is not the slam-dunk everyone sells it as. [4]
Discontinuation is common in real life, and not always because people “quit.” In a clinical-practice study using EHR data (2022–2024), the top reasons for stopping obesity pharmacotherapy with injectable semaglutide/tirzepatide within a year were cost/insurance (47.6%), side effects (14.6%), and shortages (11.8%). [5] In a claims-based real-world study of commercially insured adults without diabetes using GLP‑1s for obesity, only ~32% were persistent at one year (definition: no ≥60-day gap). [6]
The under-discussed angles that matter most on the off-ramp: (1) alcohol “drift”. Semaglutide can reduce alcohol craving/intake in some people, so stopping can quietly reintroduce calories and reward-seeking; (2) reward-salience rebound. Food can feel more compelling, not just more hunger; (3) sarcopenic regain. If you lost lean mass on the way down, regain can skew toward fat unless you actively defend muscle; (4) limits of “behavioural support”. Generic lifestyle advice often doesn’t slow regain; (5) monitoring tactics. Simple, blunt tracking (weight trend, waist, alcohol, steps, strength) can catch rebound early enough to act. [7]
Medical disclaimer: this is educational, not personal medical advice. If you’re using Ozempic for type 2 diabetes or kidney/cardiovascular risk reduction, stopping can affect glucose and risk markers, plan the off-ramp with your clinician. If pregnancy or surgery is involved, timing matters. [8]
What “stopping” semaglutide really means
Pharmacokinetics you can actually use
Semaglutide is “sticky” by design. Both Wegovy and Ozempic labels describe an elimination half-life of ~1 week and a prolonged washout: ~5–7 weeks after the last 2.4 mg Wegovy dose, and ~5 weeks after the last Ozempic dose. [1]
That pharmacology creates a predictable psychological trap: you stop injections, nothing dramatic happens for a bit, then you assume you’re “safe,” and then your appetite and “food noise” surge later, right when motivation is lowest. [1]
Sources: U.S. Food and Drug Administration prescribing information for Wegovy and Ozempic. [1]
Off ramp timeline you can play around with
This is not a promise of what you will feel because people vary, but it’s aligned with labeled clearance windows and with when rebound shows up in trials and meta-analyses. [9]
What the best evidence says happens after you stop
STEP trials: the regain story most people quote (and what they leave out)
In the STEP 1 randomized trial (68 weeks), weekly semaglutide 2.4 mg plus lifestyle intervention produced a mean weight change of –14.9% vs –2.4% with placebo. [10]
The STEP 1 extension then asked the question you’re asking: what happens when you remove the drug? Over the year after withdrawal, participants regained about two‑thirds of their prior weight loss, and improvements in blood pressure, lipids, and glycemic markers tended to revert toward baseline. [2] A commonly reported framing from that paper: by week 120, the semaglutide arm regained ~11.6 percentage points of lost weight (placebo: ~1.9), leaving net losses of ~5.6% vs ~0.1% from baseline. [2]
What most blog posts miss is the design nuance: STEP 1 extension also removed active lifestyle support during the off-treatment year, which is more “real life” (support often drops) than it is “best case.” [2] That doesn’t magically mean lifestyle doesn’t matter, but it does mean the extension reflects what happens when pharmacology ends and the scaffolding fades too.
STEP 4: a cleaner “continue vs stop” withdrawal test
STEP 4 is the blunt instrument: everyone got semaglutide during a 20-week run-in, then people were randomized to continue semaglutide vs switch to placebo (with lifestyle intervention across groups). The results were stark: from week 20 to week 68, those continuing semaglutide changed –7.9% vs +6.9% for those switched to placebo. [11]
STEP 4 also matters for expectations about side effects: among those who could tolerate titration to 2.4 mg, GI event prevalence tended to decrease over time with ongoing treatment, and switching to placebo reduced GI event reporting vs continued semaglutide during the randomised period. [12]
The big-picture meta-analyses: regain is not just a semaglutide story
A 2026 systematic review and meta-analysis in The BMJ pooled 37 studies (9,341 participants) examining weight change after stopping weight management medications. It estimated an average regain rate of ~0.4 kg/month, projecting a return to baseline weight by about 1.7 years after cessation; for incretin mimetics overall and for newer/more effective incretins, modelled regain rates were higher (up to ~0.8 kg/month), with projected return to baseline around 1.5–1.6 years. [4]
The same analysis modelled cardiometabolic markers (HbA1c, fasting glucose, blood pressure, cholesterol, triglycerides) trending back toward baseline, with projected return close to baseline by about 1.4 years after stopping. [4] The important framing: this isn’t “you failed.” It’s that the intervention ended, and chronic physiology tends to drift back.
A separate 2025 systematic review/meta-analysis focused specifically on discontinuation of GLP‑1 receptor agonists also reported “metabolic rebound”: in obesity populations, stopping was associated with significant weight gain plus deterioration in HbA1c, waist circumference, BMI, systolic blood pressure, and fasting glucose (with substantial heterogeneity across included RCTs). [13]
The contrarian truth about stopping: discontinuation is normal, not rare
In real-world settings, long-term persistence is low. A claims-based study of commercially insured adults without diabetes using GLP‑1s for weight loss found one-year persistence of ~32% overall (with product-level variation). [6]
And in a clinical-practice EHR study that actually read “why did you stop?” notes, almost half stopped due to cost/insurance, with meaningful fractions due to side effects and shortages. [5]
Why rebound happens (beyond “your appetite returns”)
Metabolic adaptation: your body gets efficient when you shrink
Long-term weight maintenance is hard partly because energy expenditure often drops more than expected after weight loss (adaptive thermogenesis), and that reduced expenditure can persist while weight is suppressed creating a biological “tailwind” for regain. [14]
This matters post-semaglutide because the drug was lowering appetite and supporting a calorie deficit. When the drug effect fades, appetite can rise into a metabolism that’s still running “efficient,” which is an ugly combo for maintenance. [14]
Adipose “memory” and catch-up fat: regain can be biased toward fat
A genuinely under-discussed research direction: adipose tissue may retain an epigenetic/transcriptional memory of obesity after weight loss, potentially shaping how fat tissue responds when energy surplus returns. This is early-stage mechanistic science (not a personal destiny), but it supports a practical point: your body may be primed to re-expand fat cells after loss, which is why environment + monitoring matters more than motivation speeches. [15]
Related concept: “catch-up fat” models suggest regain after restriction can disproportionately restore fat mass, linked with suppressed thermogenesis and high efficiency for fat deposition, again emphasising why defending lean mass changes the game. [16]
Lean mass loss: the “sarcopenic regain” problem
In the STEP 1 DEXA sub-study analysis, semaglutide 2.4 mg reduced fat mass substantially, but total lean body mass also decreased (reporting around –9.7% from baseline), even though the proportion of lean mass increased because fat mass fell more. [7]
Here’s the contrarian implication: if you come off semaglutide after losing weight but without explicitly rebuilding or defending muscle, you can regain in a “worse ratio” (more fat relative to lean), which tends to make appetite regulation, physical function, and long-term maintenance harder. Reviews on sarcopenic obesity increasingly flag pharmacologically induced weight loss as a context where muscle health needs active protection, not just passive hope. [17]
GI and gastric emptying: it’s more nuanced than “it slows your stomach”
Semaglutide is associated with significant gastrointestinal adverse reactions (nausea, vomiting, diarrhoea, constipation, abdominal pain), and Wegovy labelling reports GI adverse reactions in adult weight-loss trials in ~73% of Wegovy-treated patients vs ~47% with placebo; nausea, vomiting, and diarrhoea were among the most frequent. [9]
But gastric emptying is not a one-note story. Ozempic labelling notes semaglutide delays early postprandial gastric emptying. Yet a dedicated trial in adults with obesity found no evidence of delayed gastric emptying at week 20 for semaglutide 2.4 mg when assessed indirectly via paracetamol absorption while still showing reduced appetite, reduced cravings, and lower energy intake. [19]
Translation: some of what people experience as “GLP‑1 fullness” is likely central appetite/reward modulation, not just a slow stomach. [19]
The under-discussed angles that actually change your off-ramp
Alcohol rebound: the calorie source you didn’t know the drug was muting
There’s now randomised trial evidence that semaglutide can reduce alcohol consumption and craving in adults with alcohol use disorder (phase 2 RCT, 9 weeks), building on a broader mechanistic literature suggesting GLP‑1 receptor agonists can reduce alcohol reward and intake. [20]
So here’s the contrarian, practical warning: some of your weight loss on semaglutide may have come from accidentally drinking less (or caring less about alcohol). When you stop, alcohol reward can creep back in, adding calories, degrading sleep, increasing snack drive, and amplifying rebound. This is rarely addressed in “Ozempic withdrawal” content, but it’s an actionable variable you can monitor. [20]
Monitoring tactic: track alcohol (days/week and drinks/day) for at least the first 8–12 weeks after the last dose the same way you’d track weight. [20]
Reward-salience rebound: food can feel louder (even if protein is “fine”)
Semaglutide affects appetite control and reduces cravings and ad libitum energy intake, with evidence that it also improves “control of eating.” [19] More broadly, reviews describe GLP‑1 receptor agonists as modulators of reward dysfunction linked to food cues, potentially via dopaminergic signalling pathways. [21]
What that means on the off-ramp: you may not just feel hungrier, you may feel that food is more rewarding again. If your entire maintenance plan is “high protein + fibre,” you can still be blindsided by reward-driven eating when the drug’s reward-dampening effects lift. [21]
Practical reframing: treat “food noise” as a symptom you can design around, not a character flaw. Environmental friction (default meals, fewer trigger foods at home, earlier bedtime, fewer alcohol cues) can outperform motivation in this phase. [22]
Sarcopenic regain: why your scale “maintenance” might still be a loss
Even outside GLP‑1s, weight cycling is discussed as a potential risk for sarcopenia and adverse body-composition trajectories, especially with aging. [23] Combine that with evidence of lean mass reductions during semaglutide weight loss, and you get a real risk: you regain weight but not strength, ending up softer, weaker, and metabolically less resilient even if the scale “isn’t that bad.” [7]
That’s why resistance training and protein aren’t gym-bro extras here; they’re defensive infrastructure. [24]
The uncomfortable evidence about behavioural support
In the 2026 BMJ meta-analysis, researchers found no evidence that the intensity of behavioural support during medication treatment changed the rate of regain after cessation and no evidence that providing behavioural weight management support after stopping altered regain rates (given limitations and available data). [4]
This matters because internet advice often implies: “Use the drug to build habits, then you’ll be fine.” That might be true for some individuals but it is not the generalisable, evidence-backed guarantee people sell. [4]
The more hopeful nuance: a randomised study with post-treatment follow-up found that exercise (especially when combined with GLP‑1 therapy) improved maintenance outcomes after medication ended compared with liraglutide alone, pointing to exercise as a potentially special lever. [25]
Tapering: promising, but (still) not proven the way TikTok implies
Because semaglutide has a long half-life, your body experiences a gradual decline in exposure even if you stop injections abruptly. [1] That’s one reason formal “tapering” is not biologically required in the same way it is for dependence-forming drugs.
But people taper for other reasons, anxiety, side effect management, or trying to blunt rebound. The best publicly available tapering data right now is not a randomised trial; it’s real-world cohort evidence presented as a conference abstract from a Danish digital program, where participants who tapered had relatively stable weight at 26 weeks post-cessation (with caveats, follow-up limits, and restart rates). [26]
So the contrarian stance is both/and:
Dismiss the hype (“tapering solves rebound”) because we do not yet have strong causal evidence.
Don’t dismiss the signal either: tapering + structured support may help some people, and it’s reasonable to discuss as a strategy—especially when the alternative is an unplanned stop due to cost/shortage. [26]
The off-ramp playbook: practical, evidence-backed ways to reduce rebound
This section is written like a plan you can operationalise. None of it is magic. It’s about stacking small advantages during the 5–7 week clearance window and the 8–12 weeks after.
Start before the last dose if you can
If you’re stopping because of a planned life event (cost change, pregnancy planning, surgery timing), use the long pharmacokinetic “tail” as training wheels: your hunger is coming back gradually, so you can build routines while the drug still buffers you. [1]
If you’re stopping because of side effects, you may not have this runway, and that’s okay. But if you do have it, it’s the highest-leverage time to install defaults.
Build a “satiety stack” that survives without semaglutide
Protein is muscle defense + appetite help. A 2024 systematic review/meta-analysis of trials in adults with overweight/obesity found increased protein intake significantly prevented muscle mass decline during weight loss interventions (effects on strength/function were less consistent). [27]
Fibre supports satiety (though effects vary). A systematic review on fibre and satiety/energy intake notes that certain fibre types and doses can reduce appetite and energy intake, supporting the general strategy of higher-volume, higher-fibre eating when hunger rebounds. [28]
A practical “satiety stack” meal template:
A clear protein anchor,
A high-fibre volume component (vegetables, legumes, whole grains if tolerated),
A controlled-energy “fat/fun” component so you don’t feel punished.
This is not perfect nutrition; it’s maintenance architecture. [27]
Treat resistance training like a prescription, not a hobby
If you do nothing else, do this: lift (or do progressive resistance work) consistently. A 2025 systematic review/meta-analysis in BMJ Open Sport & Exercise Medicine reported resistance training improves fat-free mass preservation (moderate certainty) and reduces fat mass (high certainty) in weight loss contexts. [24]
Your off-ramp goal is not “exercise more.” It’s:
Maintain or grow strength (a proxy for lean mass/function),
Which helps protect against sarcopenic regain and makes daily activity easier. [17]
If you want an evidence-based mindset: semaglutide helped change energy intake; resistance training helps protect what you’re made of while energy intake changes. [7]
Exercise might be the one lifestyle lever that actually moves post-GLP‑1 outcomes
The BMJ 2026 meta-analysis didn’t find behavioural support intensity changed regain rates overall. [4] But a randomised study with one-year post-treatment follow-up found that combining GLP‑1 therapy with exercise produced better maintenance outcomes after treatment ended compared with GLP‑1 therapy alone (this was liraglutide, not semaglutide, but it’s still highly relevant to GLP‑1 off-ramps). [25]
If you’re building an “anti-rebound budget,” exercise is a better place to spend effort than obsessing over micro-optimising meal timing.
Monitor the variables that drive rebound (not just the scale)
Self-weighing is one of the least glamorous, most effective maintenance tools. Reviews and trials generally show more frequent self-weighing is associated with better weight control and can prevent some regain when used as feedback. [29]
A monitoring set that fits in real life:
Weight trend (e.g., 3–7 day rolling average),
Waist circumference (weekly),
Alcohol (days and drinks),
Steps or activity minutes,
A simple strength metric (e.g., how many push-ups, or your main lifts).
This is also aligned with behaviors observed among long-term maintainers in registries like the National Weight Control Registry (high activity + consistent self-monitoring patterns are common). [30]
Pregnancy and surgery: the non-negotiable timing issues
Pregnancy planning: Both Wegovy and Ozempic labelling instruct discontinuation at least 2 months before a planned pregnancy due to the long washout period for semaglutide. [1]
Surgery/anaesthesia: Both labels include warnings about rare post-marketing reports of pulmonary aspiration during general anaesthesia or deep sedation, related to delayed gastric emptying and residual gastric contents despite fasting; the labels explicitly instruct patients to inform providers of planned surgeries/procedures. [31]
Guidance is evolving. A 2024 multi-society statement (including the American Society of Anaesthesiologists) suggested most patients can continue GLP‑1 receptor agonists before elective surgery, with risk-stratified approaches for those at higher GI risk. [32] Earlier ASA guidance in 2023 was more conservative about holding doses, which is a good reminder that the peri-operative story is actively being refined. [33]
FAQ
-
Not in the classic drug-dependence sense. But many people experience rebound effects, especially appetite return and weight regain, because the appetite/reward modulation from semaglutide fades as the drug clears over ~5–7 weeks (Wegovy) or ~5 weeks (Ozempic). [9]
-
Semaglutide’s half-life is ~1 week. Wegovy labelling notes presence in circulation for about 5–7 weeks after the last 2.4 mg dose; Ozempic labelling notes about 5 weeks after the last dose. [1]
-
In the STEP 1 extension, participants regained about two-thirds of their prior weight loss over the year after stopping semaglutide 2.4 mg. A 2026 BMJ meta-analysis estimated average regain around 0.4 kg/month after stopping weight management meds, with faster regain after newer incretin drugs. [2]
-
There’s no high-quality evidence that tapering is necessary for safety (semaglutide clears gradually due to its long half-life). Tapering to blunt rebound is plausible but not definitively proven; real-world cohort evidence suggests some people maintain weight short-term with tapering plus structured support, but it’s not randomised causal evidence. [1]
-
Two big ones: loss of lean mass during weight loss (making regain skew toward fat) and reward-salience shifts (food and alcohol can feel more compelling again). Both can increase energy intake without you noticing. [7]
-
Weight trend and waist are useful, but also track alcohol intake, steps/activity, and a simple strength marker. Frequent self-weighing is associated with better weight control and can prevent some regain by providing early feedback. [29]
-
Both Wegovy and Ozempic labelling recommend stopping at least 2 months before a planned pregnancy due to the long washout. [1]
-
Tell your surgical/anaesthesia team you’re taking (or recently took) semaglutide. Labels warn of rare aspiration reports during anaesthesia/deep sedation, and multi-society guidance recommends a risk-stratified approach. [8]
Prioritised reference list
Here are some of the important references from this article. All references are available on request.
1. Wegovy prescribing information (PK clearance, pregnancy, surgery, adverse reactions). https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215256s024lbl.pdf
3. STEP 4 withdrawal trial (JAMA 2021). https://jamanetwork.com/journals/jama/fullarticle/2777886
4. The BMJ 2026 systematic review/meta-analysis on post-cessation regain and behavioural-support limits. https://pmc.ncbi.nlm.nih.gov/articles/PMC12776922/
5. Real-world discontinuation reasons (EHR study). https://pmc.ncbi.nlm.nih.gov/articles/PMC12636059/
6. Real-world persistence study (claims-based). https://pmc.ncbi.nlm.nih.gov/articles/PMC11293763/
7. STEP 1 DEXA substudy (lean mass changes). https://pmc.ncbi.nlm.nih.gov/articles/PMC8089287/
10. STEP 1 (NEJM 2021) and STEP 1 extension (full text). https://www.nejm.org/doi/full/10.1056/NEJMoa2032183
20. Semaglutide and alcohol use disorder RCT + mechanistic reviews. https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2829811
31. Ozempic prescribing information (PK clearance, CKD/CV indications, surgery warning, pregnancy). https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl.pdf
32. Multi-society perioperative guidance (2024). https://www.asahq.org/about-asa/newsroom/news-releases/2024/10/new-multi-society-glp-1-guidance